Macrophages, which originate from the hepatic microenvironment and are the main cellular components of the liver, are cells with high adaptation capacity and are rapidly affected by the deterioration of hepatic homeostasis. They play an important role in acute and chronic liver diseases, injury and repair processes. Macrophages, whose main functions are the production of phagocytosis and inflammatory mediators, and whose basic activities are controlled by surface receptors, are involved both in physiological processes and in various pathological conditions such as infection, inflammation, cancer and atherosclerosis
9-11. There are two distinct macrophage polarization phenotypes, which are classically activated M1 macrophages and alternatively activated M2 macrophages
12-15. M1 macrophages; They are CD68 positive, stimulated mainly by IFN-ɣ, and have high phagocytosis ability and cytotoxicity. M2 type macrophages are CD163 positive and CD204 positive, and they are mainly stimulated by IL-4 and serve in reparative fibrosis
16.
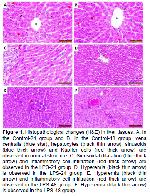
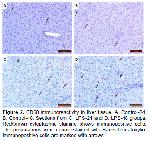
LPS, one of the components of gram-negative bacteria, is a dangerous endotoxin that strongly induces cytokine release in the inflammatory response in the host 17. The elimination of LPS in the liver is mediated mainly by Kupffer cells 18. LPS stimulates Kupffer cells via TLR4 signaling to release proinflammatory cytokines such as TNF-α and IL-1 19-21. Activation of TLRs has been reported to induce M1 polarized macrophage response inducing proinflammatory activation 22. TLR4 activation is thought to cause cellular hyperactivation and accumulation of neutrophils in narrow capillaries of the liver 23. In an experimental study in rats, it was reported that LPS administration caused inflammatory cell infiltration 24. In our study, hyperemia and inflammatory cell infiltration were observed in rat liver tissue after LPS stimulation. We think that these histopathological changes in the liver tissue may be related to the above-mentioned mechanism. In our study, it was determined that CD68 positive M1 macrophages in the rat liver after LPS stimulation were statistically significantly higher when compared to the control groups. Moreover, it was determined that CD68 immunoreactivity was significantly increased in the LPS-48 group compared to the LPS-24 group. This significant and sustained increase is probably due to activation of CD68 positive M1 macrophage cells with high phagocytosis ability and cytotoxicity via TLR 4 signaling. Thus, elimination of LPS, a dangerous endotoxin, and tissue defense will be facilitated.
In a study with mouse bone marrow cell cultures from the femur, it was shown that macrophages that were not induced by immunohistochemical staining had M1 and M2 profiles. As a result of the application of LPS to in vitro mouse cultures, it was determined that the macrophages were polarized to M1 macrophages. Uninduced macrophages show a low M1 and M2 profile, while macrophages show a high M1/M2 ratio when induced by LPS. The findings of this study are highly similar to our study, in which the number of M1 macrophages increased after induction with LPS 25. In another study, it was determined that macrophage immunophenotypes showed different distribution and kinetics in cholestatic rat liver lesions induced by alpha naphthyl isothiocyanate. It was observed that CD163 positive macrophages increased in the early period, while CD204 and CD68 positive macrophages increased continuously throughout the experiment 26. In a study conducted in cattle liver with Fasciola hepatica infection, it was found that CD68 and CD163 immunopositive macrophages increased and CD204 immunopositive macrophages decreased in fibrotic livers compared to control livers 27. Macrophages, whose main activities are controlled by surface receptors, including phagocytosis and production of inflammatory mediators, also play an important role in various pathological conditions such as inflammation, infection, atherosclerosis and cancer 28,29.
Heterogeneity has been reported to be one of the most important features of macrophages. In different diseases, macrophages can be polarized to different phenotypes. For example, in most tumors, macrophages are thought to be polarized to the M2 phenotype 30.
It has been reported that M1-like macrophages are more dominant in the early stages of inflammatory responses, while M2-like macrophages are more frequently associated with the chronic inflammation process 31-33.
It is thought that M1 macrophages are responsible for resistance against Listeria monocytogenes and Salmonella infections, generally together with intracellular pathogens 34,35. When the effect of new treatments for acute inflammation was investigated, it was determined that systemic inflammation induced by LPS was the best model 36. Therefore, in our study, we aimed to determine the M1/M2 macrophage polarization in the liver in systemic inflammation induced by LPS, and to reveal it by immunohistochemical methods.
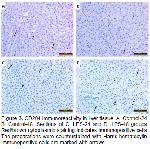
In conclusion, this study revealed the immunohistochemical distribution of M1 and M2 macrophages in vivo in rat liver at 24 and 48 hours after LPS stimulation. CD68 immunoreactivity with an increase in M1 polarization in vivo in rat liver at 24 and 48 hours after LPS stimulation; It was determined that it was statistically significantly higher when compared to the control groups. On the other hand, CD204 and CD163 immunoreactivity, which determines M2 polarization; When compared to the control groups, no change was observed in the LPS-24 group, while an increase was determined in the LPS-48 group, but this increase was not statistically significant. It was determined that after antigenic stimulation with lipopolysaccharide, rat hepatic macrophage phenotypes increased in the direction of M1 polarization, which has high phagocytosis ability and cytotoxicity. We believe that the information obtained as a result of this study may also be useful for the examination of experimentally created rat liver lesions.