Dead tissue covering the burn wound also serves as a medium for bacterial growth, reduces the host's resistance to infection, and delays the formation of granulation tissue and the re-epithelialization. Therefore, wound debridement is an imperative to the healing of burns. Surgical tangential excision of dead tissue, the repeated application of moistened dressings, hydrocolloid or semiocclusive dressings, dextranomers, intracavity gels, hydrosurgery, or various enzyme preparations can be used of burn debridement
4,6,7,23.
While effective, surgical tangential excision has several major disadvantages. This method is non-selective and it is technically difficult to control the amount of tissue to be removed, often converting a partial thickness burn into a full thickness defect. In addition, tangential excision may be associated with pain and bleeding1,5,6,12.
Different test animals such as pig6,24, Guinea pig5,13 and rat14,19,22 were used for various studies. We used rat model in the present study similar to some investigators14,19,22. The model used here is simple and repeatable. The burned wound healing model provides an in vivo approach for studying the healing of burned wounds in domestic animals.
Topical agents with benefits only as antimicrobials include silver nitrate, sulfamylon and a combination of a sulfonamide and SSD. Sulfamylon has wide spectrum activity, but it is easily absorbed systemically and can result in toxic complications. SSD has become the standard topical treatment for burn wounds17. Thus, we have chosen SSD in our study.
A wide range of enzymatic debridement have been used in the treatment of burn injury both clinically and experimentally. Among them; trypsin5, bromelain1,5, collagenase2,3,13, krill enzymes6, and papain11,14 are advocated. These agents offer the advantage of easy application with additionally being relatively safe and effective12.
Collagenase is an essential component in the wound healing process. Collagenase is best used for digesting collagen and elastin, but it does not degrade fibrin. This enzyme is responsible for the breakdown of collagen and its presence is vital to maintain the dynamic equilibrium between the breakdown of old collagen and new collagen formation in the scar-remodeling phase16. This agent should be applied only to the nonviable tissue within the wound and not to the surrounding normal skin or tissue12. Therefore, in this study, collagenase ointment was applicated only on the burned tissue.
Detergents, soaps, antiseptic solutions, and heavy metal ions are decreased efficacy of collagenase8. When using enzymatic debridement, the main problem is infection as is reported by Klasen15 and Özcan et al.2. In the present study topical antiseptic solutions and antimicrobial agent were not used with collagenase, and no infections were observed.
Alaçam et al.10 treated chronic wounds located between udder and hind extremities using collagenase in cows. They reported that collagenase had a beneficial effects on the healing of these wounds. The results of current study showed that collagenase ointment plays a role in healing of burned skin wounds possible via its enzymatic debridement effect.
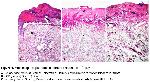
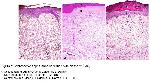
The present study showed that early nonsurgical removal of injured tissues is an effective treatment for thermal burns. Collagenase debridement accelerated burn wound healing as determined by the rate of lesion area closure and histopathological evidence. Histopathological comparison of the three groups indicated that healing of burned skin wounds was best in the collagenase group (p<0.0001).
Consequently, the data and observations collected in this study indicated that collagenase could be applied to treatment of thermal burns and it may be viable and desirable alternative to the use of SSD. Acknowledging the limitations of rat model of this experimental animal study, we believe that further studies are warranted to investigate whether collagenase is useful in domestic animals with burn wounds.